ABDR Data Managers Julia, Marina, Linda and Deirdre spoke to HFA about their role with Australian Bleeding Disorders Registry (ABDR) data – and had a lot to say about why the ABDR and MyABDR are an important part of the life of the patient with a bleeding disorder.
JULIA EKERT, MARINA GORUPPI, LINDA MASON AND DEIRDRE TUCK
Julia Ekert is Haemophilia Data/Product Manager at the Department of Clinical Haematology, The Royal Children's Hospital, Melbourne, Victoria.
Marina Goruppi is Australian Bleeding Disorders Registry (ABDR) Data Manager at Fiona Stanley Hospital and Perth Children’s Hospital in Perth, Western Australia.
Linda Mason is Transfusion Scientist/ABDR Data Manager Qld at the Blood Bank/Haemophilia Treatment Centre at the Royal Brisbane and Women’s Hospital, Queensland.
Deirdre Tuck is Data Manager/Nurse at The Royal Hobart Hospital, Tasmania.
‘The ABDR tells the story of a patient, their bleeds and their treatment over their lifetime.’
Recording accurate and consistent information in the Australian Bleeding Disorders Registry is crucial to patient treatment care and Data Managers work as part of Haemophilia Treatment Centre (HTC) teams to ensure this takes place nationwide.
Jul
Data Manager Julia Ekert at work
Where would you find a Data Manager? There are Data Managers in every Australian state and territory. Some work full-time, while others work part-time, depending on the number of patients in their HTC. Their workplace varies. Some are based in the clinic, others in the laboratory, some work from more than one hospital, and all can work from home in a hybrid model.
The Data Managers come from a range of backgrounds – nursing, laboratory scientists and administrative streams. Their positions are administered by Australian Haemophilia Centre Directors’ Organisation (AHCDO) and supported by the National Blood Authority (NBA).
No matter where they are based, the Data Managers liaise closely and meet regularly with the Director of the HTC, Haemophilia Clinical Nurses, and other members of the multidisciplinary team.
‘I work in a laboratory in an office with no windows but I share it with the HTC Director and it’s a very collegial atmosphere in the laboratory.’
‘I am in a laboratory as well, in an open office, and I work in the Haematology Department, very much part of the wider haematology team, not just the haemophilia team.’
‘I work in an office within the HTC in both the adult and children’s HTCs. I see some of the patients at the adult Centre and chat to them while I am doing other things, such as taking blood.’

The Australian Bleeding Disorders Registry (ABDR) is the system used by HTCs around Australia for the clinical care of their patients. It is much more than a simple registry of diagnosis, with data about a patient’s bleeds, treatments and the treatment plan, results of tests and other measures, hospital admissions and related clinical interventions such as surgery, and information about treatment outcomes. It also includes details about ordering, supply and use of treatment products for each individual patient.
In these days of home treatment, it is important to know what is going on outside the hospital. Since 2014 people with bleeding disorders or parents/caregivers have been able to use the app MyABDR on their mobile device or their computer to contribute data about their or their child’s bleeds and treatment, along with a record of treatment stock they are managing at home.
The ABDR has evolved enormously since it was first established in 1988. Originally it was an Access database funded by Haemophilia Foundation Australia which was updated using spreadsheets provided by each HTC. In 2008, funding was provided by the NBA, and the ABDR became a national and very complex internet-based database. The role of the Data Manager was developed to ensure that accurate ongoing information was recorded nationwide and to co-ordinate the protocols for entering data into the ABDR.
‘We are haemophilia detectives.’

Data Manager Deirdre Tuck at work
The goal is to ensure that all the relevant information about each patient enrolled in the ABDR and MyABDR is accurately recorded in a timely manner.
Adding data to the ABDR is a complex task. A Data Manager’s work can be like being a detective, proactively investigating and building a record from pieces of information held in different hospital files to be an accurate picture of what has happened for a patient.
Each individual patient’s record will follow their treatment and care history over their lifetime and it is important that the data is correct.
It is a legal requirement that each patient provides consent to being on ABDR/MyABDR. When they have consented, the Data Manager completes all sections of information within the ABDR database. This provides a very valuable Patient Summary display in the ABDR for the treating team: a concise snapshot with a comprehensive and current overview of each patient’s diagnosis, genetic information, treatment, and health outcomes. This information can be also be discussed or shared with the patient, for example, in a clinical review, in MyABDR or an ABDR patient card.
Then ongoing information needs to be recorded: bleeds, treatment product use, relevant interactions with the health system, laboratory testing, radiology investigations – including inhibitor blood tests and adverse reactions. Hospital attendance such as Haemophilia Clinic Reviews, Emergency Department attendances, hospital admissions and surgical details at HTC or other hospitals are documented.
As hardcopy documentation is being phased out in healthcare around Australia, the use of digital medical records makes the work of the Data Manager easier and less ‘detective’ work is required. When patients move interstate, permission for access to information is transferred via ABDR so that there is a seamless transition for patient care.
Checking and recording treatment product usage is an important responsibility of Data Managers. Home delivery information, Community Pharmacy dispensing of emicizumab (Hemlibra®) and other products, travel supplies and inpatient usage are all cross-checked and recorded. If you manage your inventory of treatment product at home, imagine monitoring a state-wide treatment product inventory! This careful work is essential to ensure treatment product is available, particularly for patients residing in rural areas, and to minimise wastage of precious supplies. It also enables accurate financial reconciliation for treatment products by the NBA.

Data Manager Marina Goruppi at work
Putting this information together and checking it involves paying close attention to the work of the HTC, accessing multiple systems and scrupulous attention to detail.
‘As a Data Manager you have to work out how your system works and how you will get your information because it works differently in every hospital and in every state. You get the data in many and various ways. When there’s a clinic on and when there is a surgery you know you will have data to enter.’
‘You are part of the team. We are sitting in the office space and we can hear that there is a clinic on, or we attend the clinic, or there are various meetings for example, at my HTC we have a clinical huddle every morning.’
‘I can login to the digital medical record to verify details, for example, when people have attended the Emergency Department, or follow up on admissions or discharges. Because I am in Pathology, I have access to all of the information of every product that has been dispensed that month. So at the beginning of a new month, I can go and call up all of the information on product that has been dispensed. And it has the name and the date, and then I can go into the digital medical record and find out why it was dispensed.’
‘With home deliveries, the companies have an agreement with the NBA to send a monthly report of all the deliveries to the HTCs so that we can see what product was delivered. But at the same time the product orders are checked by the HTC, the company does an inventory with the patient and then the company sends an email with the requested amount to be approved by the HTC, so we can cross-check. With Hemlibra® we now get a monthly report of the community pharmacy delivered product from the company that delivers it – when it’s delivered to the pharmacy and when it's picked up by the patient.’
This work is highly skilled and benefits from access to good technology.
‘Sometimes you have two or even three systems open at once, which you can do from your desktop. But you also might need to be logged into the hospital system at the same time and interpreting the clinical information from there to add it to the ABDR.’
‘Two screens make a big difference – sometimes three would be great!’
Health information systems require very high levels of privacy and security and this has always been at the forefront with the ABDR. The database is managed by the NBA and protected by world best practice security. Moreover, access to the database is restricted and governed by the ABDR Steering Committee. Access to the ABDR by hospital staff is limited to specific health professional roles, such as the HTC team and Data Managers, and must be approved by the HTC Director. Patient privacy is taken very seriously and guarded carefully.
‘We can look at hospital data but hospitals can’t look at ABDR data. I always lock my ABDR screen when I am not near it, but one of the safeguards is that it closes down if you don’t use it for 30 minutes. Also we don’t discuss patients using names; we always use their ABDR ID number, including when we are asked to share information.’
The contributions patients and parents make in MyABDR are an essential part of the ABDR patient history. In these days of personalised treatment where people with haemophilia treat at home, the data on when and how much product they used in treatment and details of bleeding episodes play an important role in developing a treatment plan that works best for the individual.
With MyABDR the patient can record quickly on their phone and can keep a record not only of details of treatments but bleeds as well – including the part of the body, so that they can see patterns emerging.
‘For patients the ABDR is such a good record of bleeding events and treatments but it needs to show an accurate summary of their progress. When they are reviewed by the doctor, the doctor can look at their record and the ABDR tells a story. And now with new treatments like Hemlibra®, being able to keep a record of the dose and weight are important clinically. It means the individual can get the right dose of the medication.’
‘Patients are a member of the HTC team in their home treatment. Haemophilia treatment is more of a home treatment than it ever was. In order to understand what’s going on at home and whether people are having their doses as per their treatment plan, it’s really important that the patients do their part and fill out their MyABDR records – so we know what’s going on at home.’
‘It can also show how effective treatments are. For example, one of our adult patients was constantly treating himself because he was always bleeding. He’s in the third year of a new treatment now and since he’s had the new treatment, he has not attended hospital. And that’s very clear in the ABDR because his dosages and no admissions are all there.’
‘People sometimes think we are “nagging” them about it, but it’s more that we want to give them the right medication, with the right treatments and the right doses. Some have now gone from treating three times a week to once a week, once a fortnight, once a month. If a patient records what they are actually doing, it really does make a big difference, especially to the nursing staff who are trying to track all the medication and work out when their next prescription is due and how things are going with the treatment.’
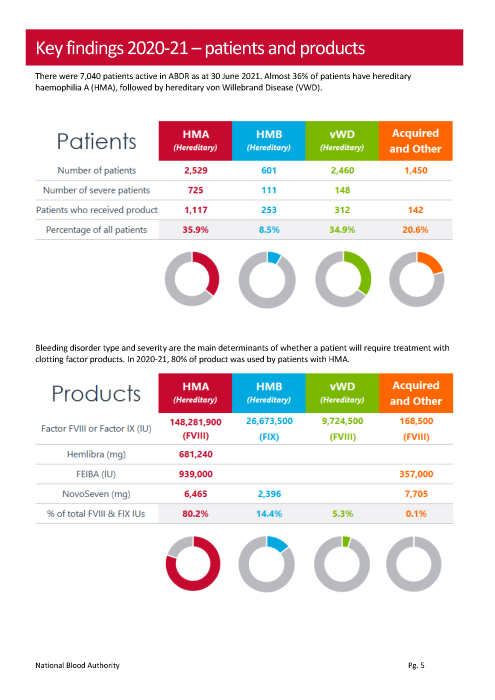
A sample of data reported in the ABDR Annual Report
The comprehensive information entered by the Data Managers enables deidentified data to be extracted from the ABDR to support research projects coordinated by AHCDO and the Annual ABDR Report, which can be seen on the NBA website. It can answer many specific questions, from how many patients have a fibrinogen disorder to what impact emicizumab/Hemlibra® has had on the factor levels of individuals with haemophilia. Research reports from the ABDR have been presented at national and international conferences and prepared for publication in peer reviewed journals.
This can provide important evidence to understand the impact of new treatments and compare them to existing treatments – what have been the outcomes for people’s quality of life, their bleeding episodes and their hospitalisations? How is this impacting on their joints?
Data from the ABDR is also central to the work of the NBA in forecasting future treatment product requirements and budgeting, essential to the ongoing supply and availability of these treatment products to Australians.
Data managers belong to the national ABDR Data Managers Group. Group members meet regularly via teleconferences and once each year meet in person (online during COVID) for study days that also coincide with HFA conference dates. Data Managers are represented on several national committees – the Research Committee, Treatment Advisory Committee and the ABDR Stakeholders Group. It is because of these opportunities that collaborative relationships have developed between the Data Managers Group, the NBA and AHCDO Members and staff, leading to a level of camaraderie that ensures the best outcomes for all.
‘I work in a very cohesive environment with the HTC Director and the haemophilia nurse and the scientists. It’s a pleasure to come to work with the group and all the latest developments are shared amongst us and we celebrate the improved quality of life of the patient.’
The Data Managers Group would like to dedicate this article to Debra Belleli (dec.), our beloved and dedicated colleague, who worked as a Data Manager at The Alfred hospital in Melbourne for many years. RIP 2022.
Haemophilia Foundation Australia acknowledges the Traditional Owners and Custodians of Country throughout Australia, the land, waters and community where we walk, live, meet and work. We pay our respects to Elders past and present and extend that respect to all Aboriginal and Torres Strait Islander peoples.
Sign up for the latest news, events and our free National Haemophilia magazine